Batteries


The lithium-ion batteries changing our lives
From smartphones and laptops to bicycles and cars, the various tools that we use on a daily basis run on electricity as energy. To improve the usability of such tools, improving the performance of batteries makes a big difference.
In that context, lithium-ion batteries have been attracting attention in recent years. Under the supervision of Ryoji Kanno, an Institute Professor at the Tokyo Institute of Technology, who has been involved in improving battery performance for more than 30 years, this series of articles explores lithium-ion batteries, from what they are to the status of research into the solid-state batteries called the next-generation lithium-ion batteries. This is a five-part series. Part 1 discusses the characteristics of lithium-ion batteries, how they generate electricity, and how they differ from lead-acid batteries.

Supervisor: Ryoji Kanno
Institute Professor (Professor Emeritus), Institute of Innovative Research, Tokyo Institute of Technology
In 1980, he completed his master’s degree in inorganic and physical chemistry at the Graduate School of Science, Osaka University. In 1985, he became a Doctor of Science. After working as an associate professor in the Faculty of Science at Kobe University, he became a professor at the Tokyo Institute of Technology Interdisciplinary Graduate School of Science and Engineering in 2001. In 2016, he became a professor at the Tokyo Institute of Technology School of Materials and Chemical Technology. In 2018, he became a professor at the Tokyo Institute of Technology Institute of Innovative Research and a leader in the All-Solid-State Battery Unit. In 2021, he became an Institute Professor at the Tokyo Institute of Technology Institute of Innovative Research and director of the Research Center for All-Solid-State Battery.
INDEX
1. What are lithium-ion batteries?
2. How do lithium-ion batteries produce electricity?
3. Are there different types of lithium-ion batteries?
4. What is the difference between lead-acid batteries and lithium-ion batteries?


Lithium-ion batteries are rechargeable batteries that are built into the smartphones and laptops that we use every day. The prototype of the battery was invented around the end of the 18th century, and batteries have evolved over more than 200 years since then. Lithium-ion batteries are one of the newest types of batteries created in the course of this evolution.
Batteries are divided into primary batteries, which can only be used once, such as dry cell batteries, and secondary batteries, which can be recharged and used many times. Lithium-ion batteries are rechargeable secondary batteries. Compared to other types of batteries, they can be made smaller and lighter, on top of which they can store large amounts of electricity.
There are various types of batteries besides lithium-ion batteries, but in fact, the basic mechanism by which they produce electricity is the same in all of them.
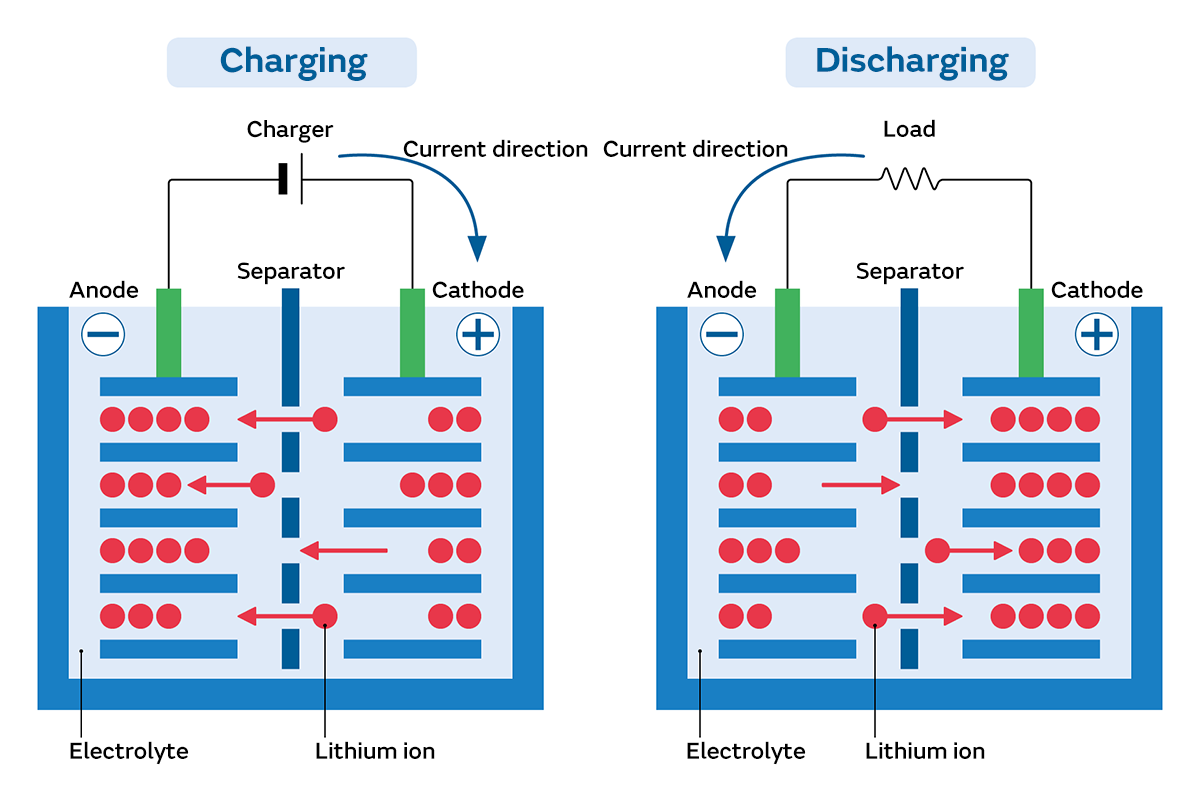
Batteries have a positive electrode (cathode) and a negative electrode (anode) made out of metal, between which they are filled with a substance (electrolyte) that conducts electricity carried by ions. The metal electrodes are dissolved by the electrolyte, dividing into ions and electrons. When the electrons move from the anode to the cathode, an electric flow (current) is generated, creating electricity. In secondary cells, electrons are stored at the anode by charging before starting to use the battery, and electricity is produced by the stored electrons moving to the cathode when using the battery.
Lithium-ion batteries use a metal compound into which lithium is embedded in advance as the cathode. Carbon, which can store that lithium, is used as the anode. This structure generates electricity without dissolving the electrodes in an electrolyte like conventional batteries. In addition to suppressing deterioration of the battery itself and allowing more electricity to be stored, this also enables the battery to be charged and discharged more times. Moreover, since lithium is a very small and light substance, it enables various advantages such as the creation of smaller and lighter batteries.
Lithium-ion batteries can be divided into several types depending on the metal used for the cathode. The first metal used for the cathode of lithium-ion batteries was cobalt. However, cobalt is a rare metal with a low output like lithium, so it has a high manufacturing cost. Now, manganese, nickel, iron, etc. have come to be used as materials that are inexpensive and have a low environmental impact. Since each material used creates a different type of lithium-ion battery, let’s look at the characteristics of each in turn.
| Type of lithium-ion battery | Voltage | Number of discharges | Pros and cons |
|---|---|---|---|
| Cobalt lithium-ion batteries | 3.7V | 500 to 1,000 |
|
| Manganese lithium-ion batteries | 3.7V | 300 to 700 |
|
| Lithium iron phosphate batteries | 3.2V | 1,000 to 2,000 |
|
| Ternary lithium-ion batteries | 3.6V | 1,000 to 2,000 |
|
Types and characteristics of lithium-ion batteries
Lithium cobalt oxide is used for the cathode. Cobalt lithium-ion batteries were the first mass-produced lithium-ion batteries because lithium cobalt oxide is relatively easy to synthesize and easy to handle. However, because cobalt is a rare metal and expensive, it is rarely used in automobile parts.
Lithium manganese oxide is used for the cathode. Manganese lithium-ion batteries can produce the same voltage as cobalt lithium-ion batteries and have the advantage that they can be made at a low cost. The disadvantage is that manganese may dissolve out into the electrolyte during charging and discharging, shortening the battery life.
Lithium iron phosphate is used for the cathode. The advantages of lithium iron phosphate batteries are that their structure does not break down easily even when heat is generated inside, they are highly safe, and they cost less to manufacture than manganese lithium-ion batteries because they use iron as a raw material. However, their voltage is lower than other lithium-ion batteries.
In order to reduce the amount of cobalt used, these batteries are made using three materials: cobalt, nickel, and manganese. Today, many of this type of battery have a higher percentage nickel. While their voltage is slightly lower than that of cobalt and manganese lithium-ion batteries, their manufacturing cost is lower. However, there are still challenges to using cobalt, nickel, and manganese as practical materials, such as difficulty in synthesizing each material and poor stability.
In addition to lithium-ion batteries, there are other types of batteries that can be recharged. Among them, lead-acid batteries have a long history of being used for more than 100 years, and even now that new batteries such as lithium-ion batteries have been developed, they continue to be used as automobile batteries.
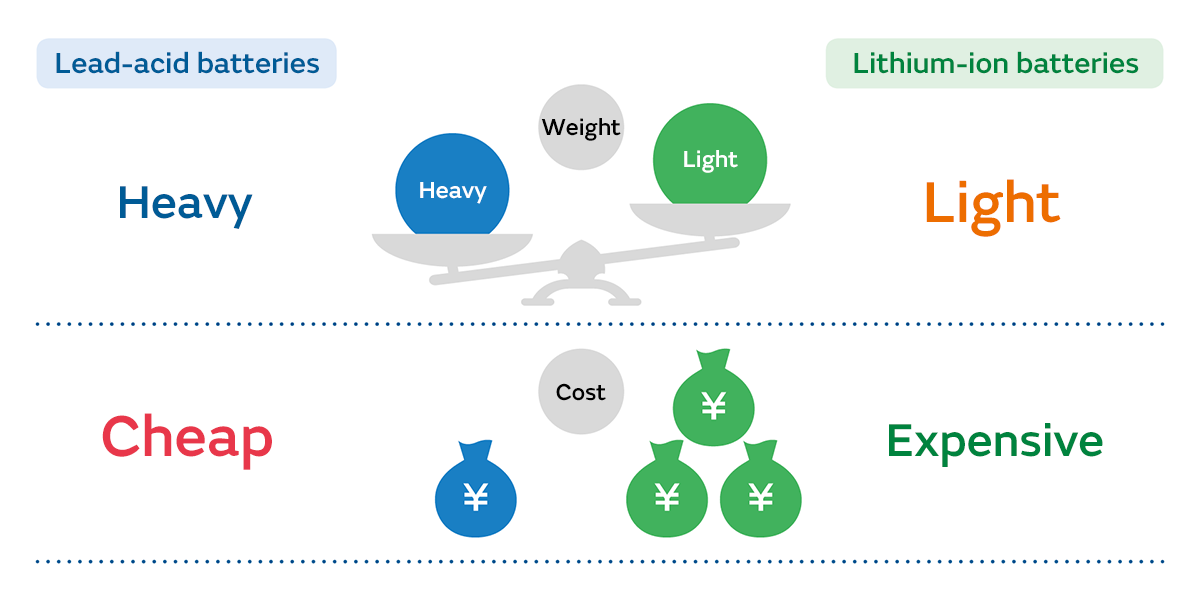
Lead-acid batteries use lead as the material for the cathode and anode, making them very inexpensive to produce compared to lithium-ion batteries. However, because lead is heavier than other metals, the batteries themselves are heavy. There are other disadvantages as well, such as the fact that the voltage can only be increased to 2 V, and self-discharge is large.
Even so, the reasons why automobile batteries are not replaced with high-performance secondary batteries such as lithium-ion batteries are that they are just inexpensive and are highly reliable batteries based on technology that is pretty much completely established at this point. Automobiles take advantage of these characteristics of lead-acid batteries, and a system has been put in place to recycle them. Trying to replace these with a new type of battery would mean having to rethink things right from the circuit design, and manufacturers don’t want to spend extra money when lead-acid batteries are doing good enough at the moment.
However, lithium-ion batteries have already been adopted as secondary batteries used to drive motors in electric vehicles and hybrid vehicles. So, it may be that lead-acid batteries are no longer used even in regular automobiles in the future.

Lithium-ion batteries, first commercialized for consumer goods in the early 1990s, were used to make video cameras smaller and lighter. After that, one manufacturer after another adopted them for mobile phones, which were expanding in popularity at the time, causing demand to spread quickly. Lithium-ion batteries are now used in various fields throughout our daily lives, including smartphones and laptops, as well as electric vehicles and electric bicycles.

The whole idea behind batteries is that they are, in a word, canned energy. Lithium-ion batteries, which store energy at a high density per unit volume, require more safety considerations than other types of batteries. Moreover, since they use a flammable organic solvent, they need to be handled more carefully than other batteries that use an aqueous solution.
The most important thing to avoid is an internal short circuit. An internal short circuit is a condition that occurs when a force applied from outside deforms the battery, causing the cathode and anode to connect directly. When the current concentrates at this connection, the temperature rises, and the battery itself ignites, which can lead to a major accident. Even the smallest impurities may cause an internal short circuit by contaminating the inside of a battery. It is therefore essential to have an accident prevention feature such as providing a protective circuit so that an excessive current does not flow in the battery.
In addition, it is important to control the temperature of the battery itself, such as by using a cooling device to keep the battery use environment at no more than 60°C. A variety of methods have been conceived to improve safety, such as having the separator, a membrane that separates the cathode from the anode, completely shut out the space between the cathode from the anode when the temperature rises above a certain level.
As explained in this article, lithium-ion batteries, which are smaller, lighter, and have higher performance than conventional secondary batteries, are likely to continue to be used throughout our lives. Part 2 looks at how lithium-ion batteries are actually used.