Batteries

The lithium-ion batteries changing our lives
Under the supervision of Ryoji Kanno, an Institute Professor at the Tokyo Institute of Technology who has been involved in improving battery performance for more than 30 years, this series of articles explores lithium-ion batteries, from what they are to the status of research into the solid-state batteries called the next-generation lithium-ion batteries. Part 2 focuses on the benefits of lithium-ion batteries and the kinds of situations where they are used in our everyday lives. It also explains how to use them so they last longer.

Supervisor: Ryoji Kanno
Institute Professor (Professor Emeritus), Institute of Innovative Research, Tokyo Institute of Technology
In 1980, he completed his master’s degree in inorganic and physical chemistry at the Graduate School of Science, Osaka University. In 1985, he became a Doctor of Science. After working as an associate professor in the Faculty of Science at Kobe University, he became a professor at the Tokyo Institute of Technology Interdisciplinary Graduate School of Science and Engineering in 2001. In 2016, he became a professor at the Tokyo Institute of Technology School of Materials and Chemical Technology. In 2018, he became a professor at the Tokyo Institute of Technology Institute of Innovative Research and a leader in the All-Solid-State Battery Unit. In 2021, he became an Institute Professor at the Tokyo Institute of Technology Institute of Innovative Research and director of the Research Center for All-Solid-State Battery.
INDEX
1. What are the benefits of using lithium-ion batteries?
2. What are the applications for lithium-ion batteries used in our everyday lives?
3. What are the applications for lithium-ion batteries used in industrial fields?
4. What are the tips and precautions for charging products that use lithium-ion batteries?
Lithium-ion batteries have many advantages in terms of safety and functionality compared to other batteries such as lead-acid batteries. The key benefits include:
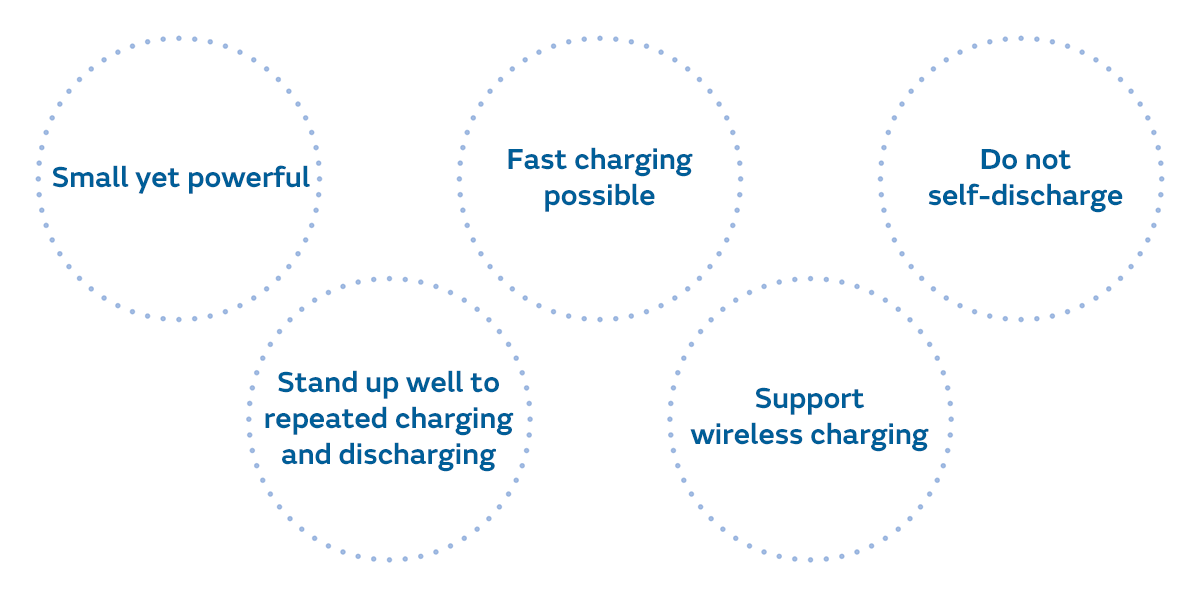
Secondary batteries that can be recharged and used repeatedly like lithium-ion batteries include nickel-metal hydride batteries and nickel-cadmium batteries in addition to lead-acid batteries. The most obvious advantage of lithium-ion batteries compared to these batteries is that they are small and light, and yet powerful.
Comparing the characteristics of these batteries at the same size, the maximum voltages they can produce are 2.1V for lead-acid batteries, 1.2V for nickel-metal hydride batteries, and 1.25V for nickel-cadmium batteries. Lithium-ion batteries, on the other hand, can produce voltages as high as 3.2 to 3.7V.
Lithium-ion batteries do not utilize chemical reactions like other secondary batteries when making electricity. Therefore, compared to other secondary batteries, their electrodes deteriorate less, allowing them to stand up very well to repeated charging and discharging.
A major feature of lithium-ion batteries is that they can be charged quickly. But fast charging, or charging done in a short time, is something that secondary batteries other than lithium-ion batteries can also do. However, with nickel-metal hydride batteries and nickel-cadmium batteries, it was difficult to determine when charging was finished, so fast charging was not put to practical use. Fast charging of lithium-ion batteries has been implemented because the charger can determine when charging is finished.
Wireless charging, or charging without a charging cable, is also possible with secondary batteries other than lithium-ion batteries, just like fast charging. However, since wireless charging technology was established in 2007, making it a relatively new technology, it was adopted for lithium-ion batteries, which were already expected to become widespread. Research is being conducted into a system that will make it possible in the future to charge electric vehicles simply by parking them in a parking lot.
Batteries display a phenomenon called “self-discharge,” the natural discharge of a battery even when it is not in use. For example, if you want to start the engine of a car that has not been driven for more than a month, you may not be able to turn the starter over because of low voltage. This so-called “dead battery” condition is caused by self-discharge.
Self-discharge occurs when a chemical reaction progresses little by little even when a battery is just left in inactive storage. This is why lithium-ion batteries, which use a slightly different reaction from the battery reaction that occurs in other secondary batteries, hardly self-discharge at all.
By the way, in the case of smartphones and PCs equipped with lithium-ion batteries, the battery may run down even when the device is not used. In this case, it is because the device is not completely turned off and is consuming a small amount of electricity even though the screen is off, so that it can start up immediately.
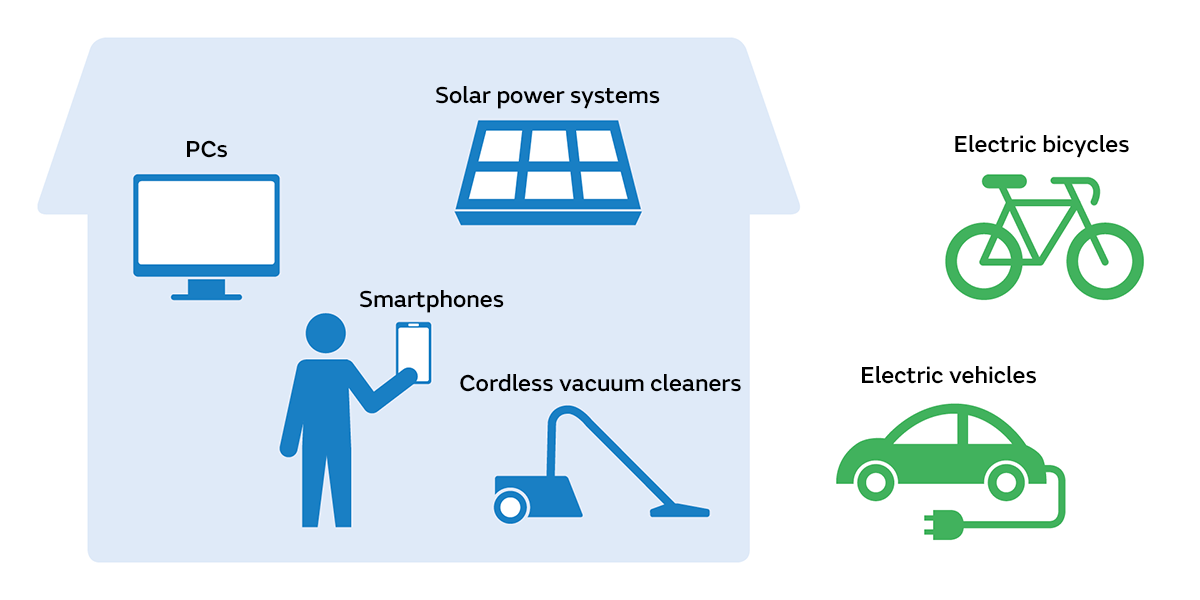
There are many tools around us that run on electricity. Taking advantage of the benefit that they are small and powerful, lithium-ion batteries are incorporated into a variety of devices. In particular, products such as smartphones, PCs, and digital cameras became smaller, lighter, and longer lasting after they started using lithium-ion batteries.
Electric vehicles were initially equipped with nickel-metal hydride batteries. However, they now use lithium-ion batteries because of their benefits in being powerful and having low self-discharge and because they do not suffer from the non-user-friendly inability to allow top-up charging like nickel-metal hydride batteries.
Lithium-ion batteries are also used for small home appliances such as cordless vacuum cleaners and irons, vehicles such as electric bicycles and electric motorcycles, and applications such as storing electricity generated during the day using a solar power generation system at home.
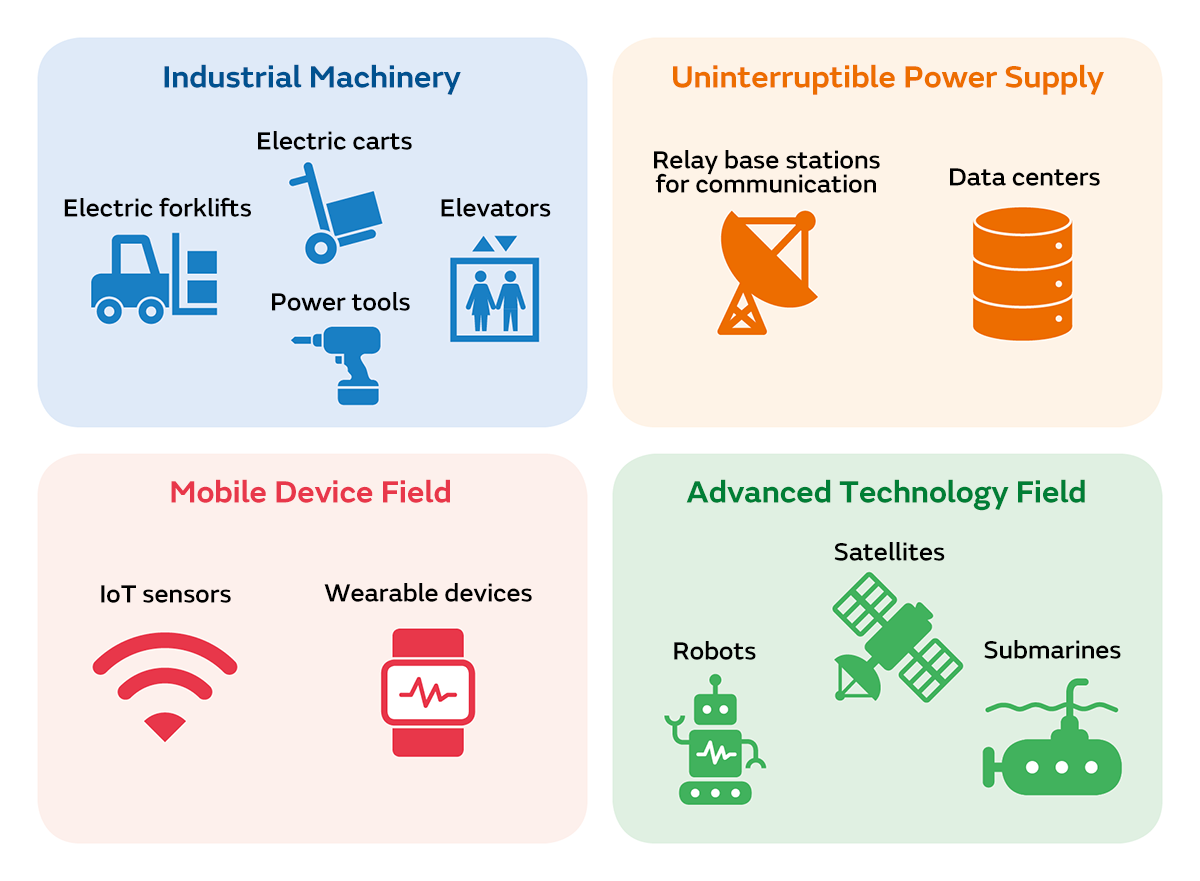
In industrial fields, lithium-ion batteries are used to cordlessly operate machines such as robots and drones. There are also a wide range of other industrial fields where lithium-ion batteries are utilized, such as for IoT sensors installed in various locations and special vehicles such as submarines and rockets.
On the other hand, lithium-ion batteries, too, have limited energy density, which limits their use when more power is required. For example, today’s lithium-ion batteries can be used as the energy source to drive the motors in electric vehicles, but it is still technologically too difficult to fly large airplanes on the energy of lithium-ion batteries. However, people are developing new vehicles such as “flying cars” that use lithium-ion batteries. There are still many possibilities left even if we just investigate thoroughly the performance potential of current lithium-ion batteries.
Just as lithium-ion batteries have different characteristics from other batteries, they will last longer if you use them with an understanding of their characteristics. Understand the points and features of charging to maintain performance.
One of the features of lithium-ion batteries is their user friendliness. For example, with nickel-metal hydride batteries and nickel-cadmium batteries, if you try to add charge while capacity remains, the battery may not appear to be charged more than that remaining capacity. This is called the “memory effect,” and it basically does not happen with lithium-ion batteries.
To prevent the memory effect in nickel-metal hydride batteries and nickel-cadmium batteries, it is recommended to charge them after using up all the electricity. Lithium-ion batteries, in contrast, can be said to be more user-friendly because you can top up their charge before running them down completely.
Devices that use lithium-ion batteries, such as smartphones and laptops, use circuits that do not allow charging beyond the battery’s capacity even if the battery is used while always charged. So, there is no worry that the battery will be overburdened, but if you want a lithium-ion battery to last longer, it is best to continue using it while charged up to about 50% and connected to a power supply. Some laptops can be set to not charge more than 50%.
Conversely, the thing to avoid with lithium-ion batteries is using a device while maintaining a 100% charged state. If you continue to use a fully charged laptop while it is connected to a power supply, it will shorten the battery life.
On the other hand, leaving a device in inactive storage in an over-discharged state where almost all the electricity is used up can also shorten the life of lithium-ion batteries. In other words, avoid use in the extreme states of being overcharged and over-discharged. This is similar to the way exercising in a state of fullness or hunger puts an undue burden on the body.
To get stable performance out of lithium-ion batteries, it is best to use them at room temperature. You may have heard the rumor that they will last longer when cooled, but this is counterproductive because the battery’s resistance value increases at low temperatures, increasing the load for charging and discharging. Moreover, cooling may cause condensation on the battery, and the circuits around the battery may short-circuit.
Lithium-ion batteries have various advantages, but it must be said that it is important to use them in ways that make the most of those advantages in order to get good performance for a long time.