Batteries

The lithium-ion batteries changing our lives
This five-part series introduces the characteristics, history, and future possibilities of lithium-ion batteries under the supervision of Ryoji Kanno, an Institute Professor at the Tokyo Institute of Technology. Part 4 focuses on solid-state batteries, which have similar characteristics to lithium-ion batteries and are said to be the “next-generation batteries.” It explains their differences from current lithium-ion batteries and examines their expected applications and the challenges to practical application.

Supervisor: Ryoji Kanno
Institute Professor (Professor Emeritus), Institute of Innovative Research, Tokyo Institute of Technology
In 1980, he completed his master’s degree in inorganic and physical chemistry at the Graduate School of Science, Osaka University. In 1985, he became a Doctor of Science. After working as an associate professor in the Faculty of Science at Kobe University, he became a professor at the Tokyo Institute of Technology Interdisciplinary Graduate School of Science and Engineering in 2001. In 2016, he became a professor at the Tokyo Institute of Technology School of Materials and Chemical Technology. In 2018, he became a professor at the Tokyo Institute of Technology Institute of Innovative Research and a leader in the All-Solid-State Battery Unit. In 2021, he became an Institute Professor at the Tokyo Institute of Technology Institute of Innovative Research and director of the Research Center for All-Solid-State Battery.
INDEX
1. What are solid-state batteries?
2. How do solid-state batteries work?
3. What are the types of solid-state batteries?
5. What are the applications of solid-state batteries?
6. How safe are solid-state batteries?
7. What are the challenges to the practical application of solid-state batteries?
As the name implies, a solid-state battery is a battery in which all the components that make up the battery are solid. Secondary batteries (batteries that can be recharged and used repeatedly) like lithium-ion batteries are basically composed of two electrodes (a cathode and an anode) made of metal and an electrolyte that fills the space between them. Conventional secondary batteries use a liquid as the electrolyte, but solid-state batteries use a solid as the electrolyte.
It is expected that the solid electrolyte will enable larger-capacity and higher-output batteries than lithium-ion batteries. Moreover, making the electrolyte solid has advantages in terms of safety over lithium-ion batteries. They are therefore attracting attention for installation in electric vehicles and other products.
In this way, it is said that solid-state batteries would have various benefits if they could be put into practical use. Currently, different companies are competing in product development and the realization of mass production for large-volume supply.
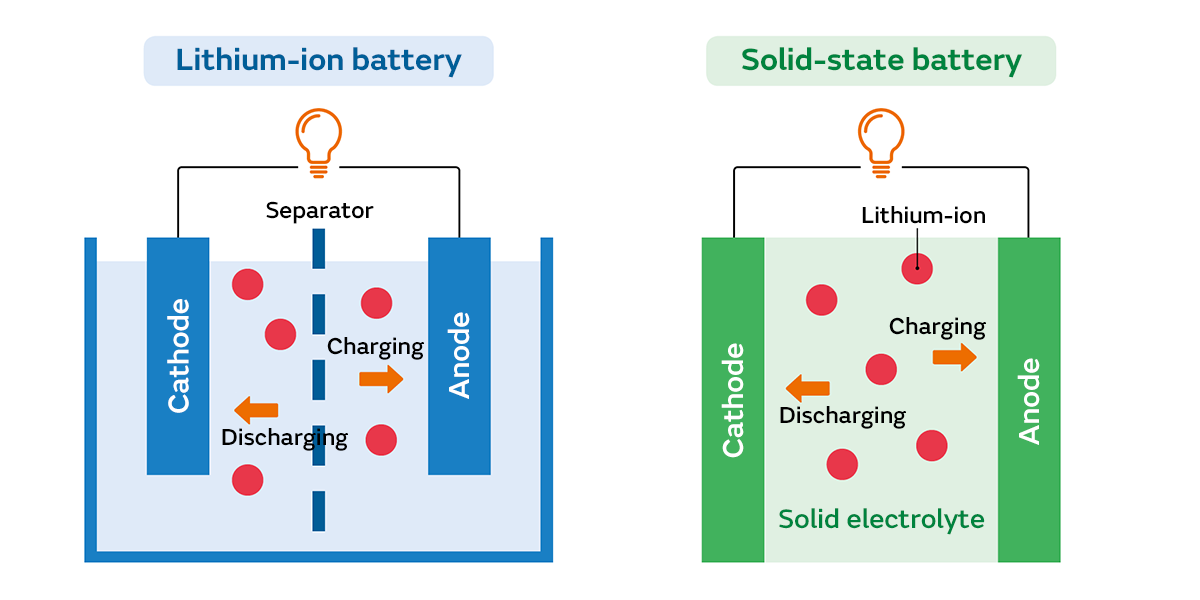
Solid-state batteries have almost the same mechanism as lithium-ion batteries for extracting electricity from the batteries. Metal is used as the material for the electrodes, and electrical flow is generated by ions moving through the electrolyte between the cathode and anode. The big difference is that the electrolyte is solid. Also, when the electrolyte is a liquid, there is a separator that separates the cathode from the anode, preventing the liquid on the cathode side from mixing suddenly with the liquid on the anode side. But in the case of a solid electrolyte, the separator is unnecessary.
The key to research into solid-state batteries is the discovery and/or development of solid-state materials. In the past, no solid-state material had been discovered that could allow ions to move around inside and create a sufficient flow of electricity to the electrodes. But the discovery of such materials has given momentum to the development of solid-state batteries. By changing from a liquid to solid electrolyte, the ions will move well in batteries, making it possible to create batteries with larger capacity and higher output than lithium-ion batteries.
Solid-state batteries are broadly classified into “bulk” and “thin-film” types depending on the manufacturing method, with the amount of energy they can store differing.
| Type | Characteristics | Anticipated uses |
|---|---|---|
| Bulk | Can store a lot of energy | Electric vehicle batteries, etc. |
| Thin-film | Can store only a small amount of energy, but lasts a long time | IoT devices, etc. |
Powders (substances consisting of powder, granular material, etc.) are used as the materials of the electrodes and electrolyte. It is possible to make large-capacity batteries that can store a lot of energy. It is anticipated that they will mainly be used for large things such as electric vehicles.
These are batteries manufactured by stacking a thin-film electrolyte on the electrodes in a vacuum state. The amount of energy stored is small and they cannot produce a large capacity. However, there are advantages such as a long cycle life and ease of manufacturing. Because they are small, they are suitable for use in small devices such as sensors.
Solid-state batteries, which are expected to be the next generation of secondary batteries, are considered to have the following benefits.
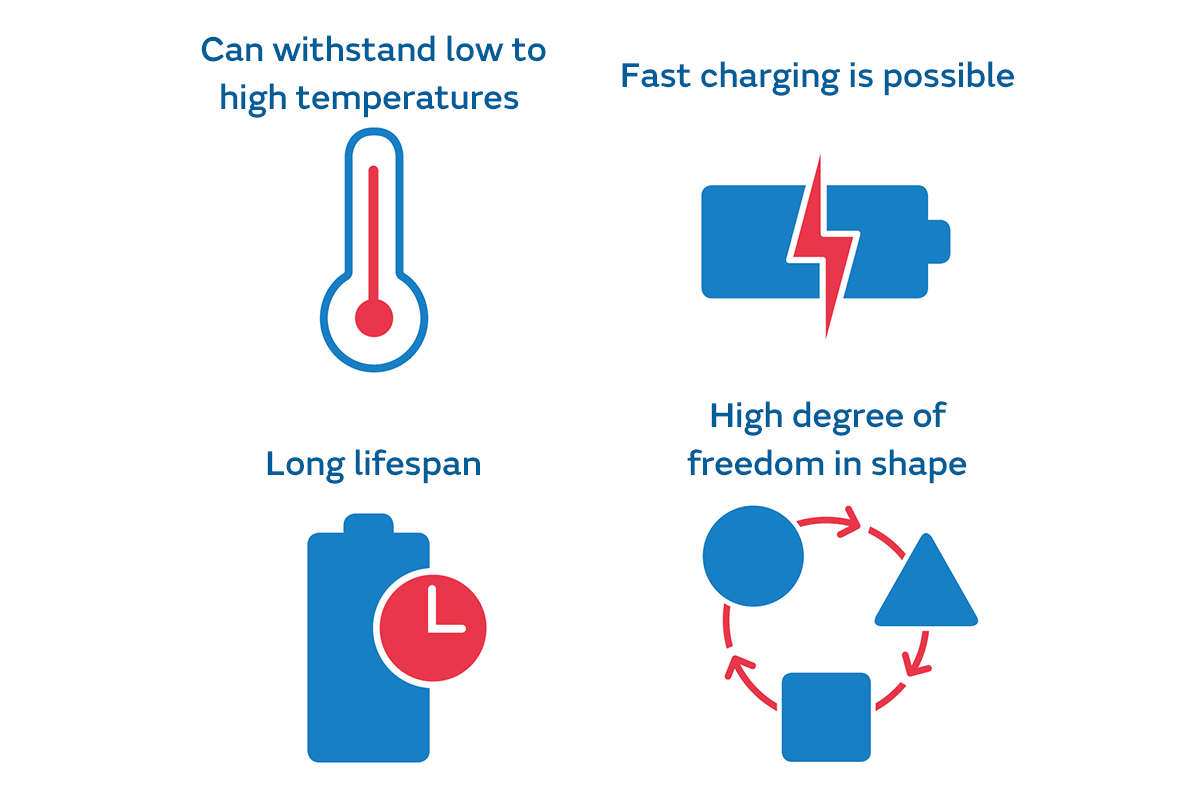
Since the electrolytes in lithium-ion batteries are made of flammable organic solvents (liquids that dissolve substances that do not dissolve in water), there is concern about their use in high-temperature environments. On the other hand, since the electrolytes in solid-state batteries are not made of flammable materials, they can be used at higher temperatures.
Further, in the case of liquids, the movement of ions slows at low temperatures, causing battery performance to drop, and the voltage may decrease. In the case of solids, the internal resistance does not increase so much and battery performance does not drop much because the solid does not freeze like a liquid even at low temperatures.
The benefit of being resistant to high heat is also advantageous for fast charging. The faster batteries charge, the more they heat up. Because of this, it is believed that it will be possible to charge high-temperature-resistant solid-state batteries even faster than current lithium-ion batteries.
The lifespan of a battery depends on the properties of the electrolyte. Since lithium-ion batteries do not use a battery reaction like other secondary batteries, the electrode deteriorates little and lasts a long time, but when used for a long time, electrolyte deterioration can be seen. In that respect, since the electrolytes in solid-state batteries deteriorate less than liquids, it will be possible to extend battery lifespan even further.
Liquid electrolytes have structural restrictions to prevent liquid leakage. But in the case of solid-state batteries, there is no such limitation. So, they can be used in various shapes because it is easy to make them smaller and thinner, and because they can be used while overlapped or bending.
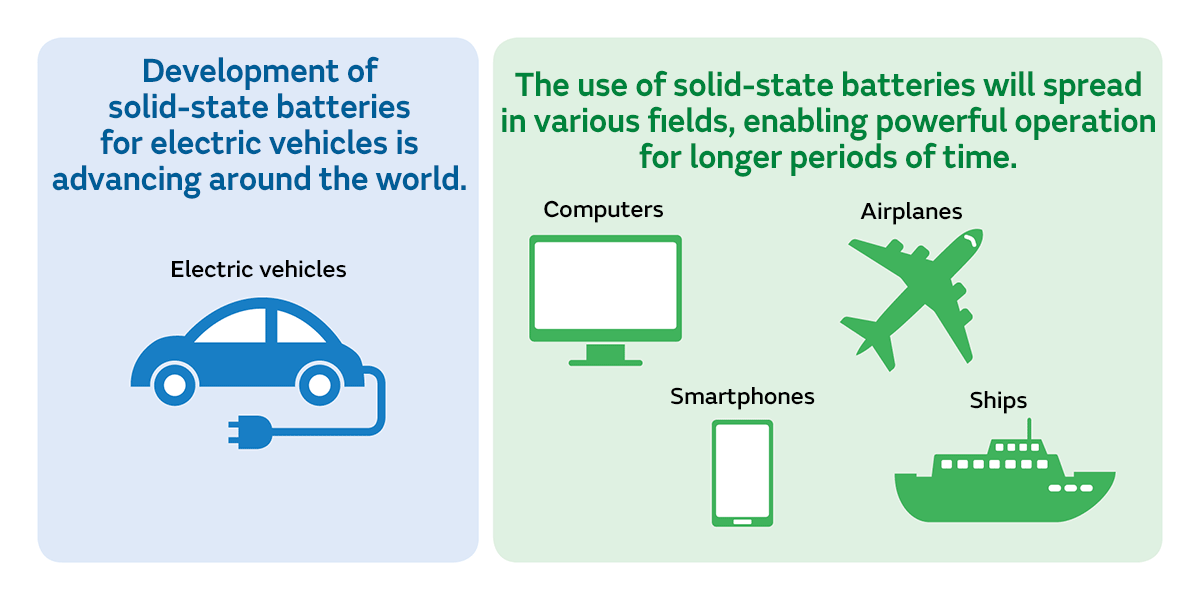
One of the expected applications for solid-state batteries is electric vehicles. Currently, electric vehicles use lithium-ion batteries. But if they used solid-state batteries, the risk of ignition due to accidents is expected to decrease since they do not contain flammable organic solvents. In addition, whereas today’s electric vehicles take longer to charge than refueling with gasoline, with solid-state batteries it will be possible to charge them more quickly.
In addition, one of the reasons why the practical application of solid-state batteries is being actively pursued is that they can compensate for lithium-ion batteries’ weak point of being vulnerable to high temperatures. Since they could be soldered directly to an electronic substrate by taking advantage of their heat-resistant characteristic, it is also anticipated that their uses will include electronic device backup power supplies and IoT sensors. If used in PCs or smartphones, they should enable powerful operation for a longer time.
Furthermore, since solid-state batteries can achieve a larger capacity and higher output than lithium-ion batteries, they can be expected to be used in airplanes and ships. And since they are resistant to temperature changes across the spectrum from high to low temperature, it can be expected that their applications will expand to include devices used in outer space.

Lithium-ion batteries use easily vaporized organic solvents as electrolytes, so there are concerns about their use in high-temperature environments. Also, in order to use liquid electrolytes, it was necessary to devise ways to keep the cathode and anode from coming into direct contact (shorting) on impact, such as by using separators between them.
Solid-state batteries are hard to short-circuit because the electrodes are separated by a solid, and they can be used at higher temperatures because they use highly heat-resistant electrolytes. However, since all batteries are “canned energy,” solid-state batteries are not risk-free. Care must be taken when handling them, as the electrodes may short-circuit for some reason.

Research and development into higher-performance solid electrolyte materials is underway with the aim of putting solid-state batteries into practical application in the early 2020s. The following challenges must also be solved to achieve this.
In order for batteries to perform well, the electrodes and electrolyte must always be in close contact. Liquid electrolytes always change shape, so they can maintain close contact even if the electrode changes a little. With solid-on-solid, on the other hand, there is the challenge that it is difficult to always maintain close contact.
In order for solid-state batteries to significantly increase energy density over existing lithium-ion batteries, it is necessary to develop electrodes that can store more power at the same weight and size.
Since the electrolyte will be changed from liquid to solid, a manufacturing process different from lithium-ion batteries is needed. For example, solid-state batteries can be based on oxides, sulfides, nitrides, etc., depending on the material. The solid electrolytes used in solid-state batteries based on sulfides, which is one of the mainstream types, are so sensitive to moisture that they degenerate even when exposed to moisture in the air. Therefore, the production of solid-state batteries, which require strict moisture control, will need dedicated facilities such as dry rooms.
As mentioned above, various companies are currently making efforts to commercialize solid-state batteries, which are expected to further enhance the performance of lithium-ion batteries. On the other hand, lithium-ion batteries are actively used in a wide range of fields. Part 5 explores how lithium-ion batteries will play a role in realizing a sustainable society.