Capacitor


Murata capacitors support every possible kind of product in society, from household electric appliances around us, such as smart phones and LED lights, to hybrid vehicles and electric vehicles, which will lead the next generation, and equipment for use in space where high reliability is required. Medical devices are, of course, no exception.
Here, we introduce our company's strengths, the concepts behind medical device products, and typical capacitors for medical devices.
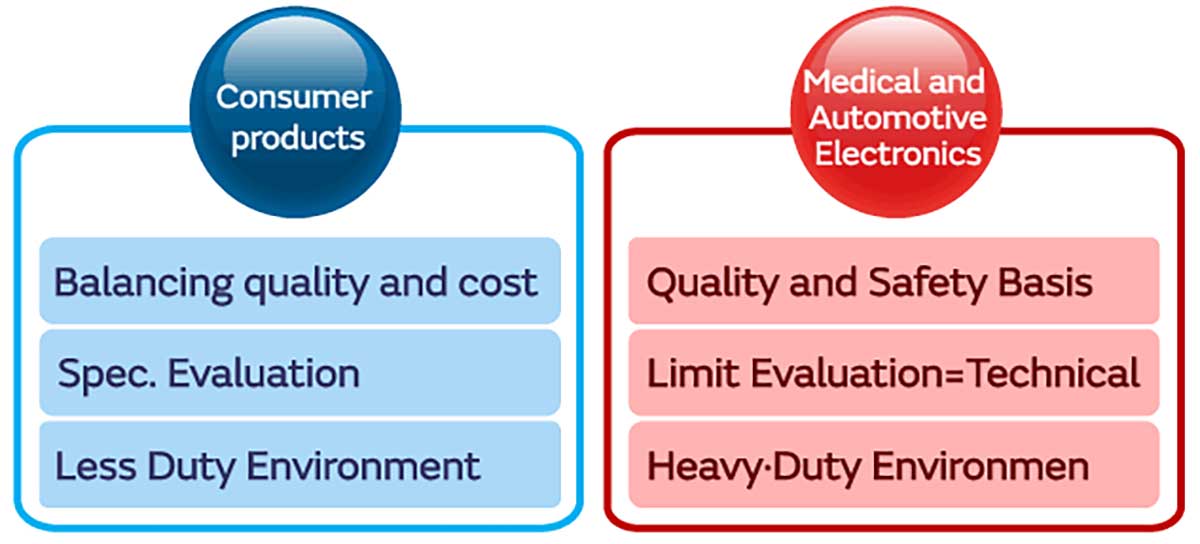
The concept and way of thinking regarding reliability for consumer devices differs completely from the concept and way of thinking for high-reliability devices such as medical devices (especially, implantable medical devices) or on-vehicle devices. With consumer devices, the trend is towards priority being given toward fixed costs, whereas with high reliability devices, considerable weight is placed on reliability, and the requirement is for zero defects. In addition, their operating environment, service life and evaluation criteria are considerably different.
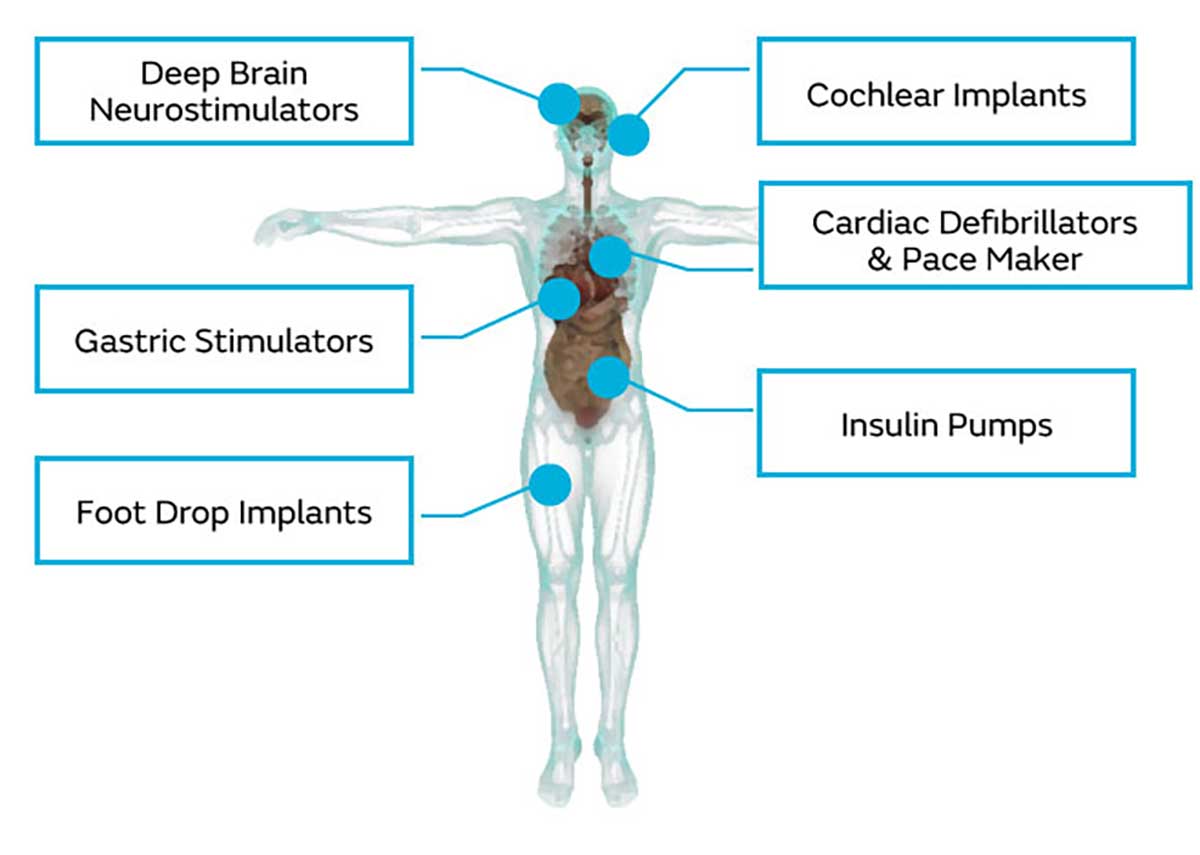
A wide lineup of characteristic Murata capacitor products is available for medical devices. Of the various medical devices available, we will be introducing typical and characteristic capacitor products for "implantable medical devices" and "portable and wearable medical devices".

Deep brain neuro-stimulators, stomach stimulators, artificial cochlea, drop foot appliances, cardioverters, pacemakers

Ultrasonic echo devices, electro cardiograms, blood gas analyzers

Because implantable medical devices, as typified by cardiac pacemakers, must be embedded in the body, a key issue is to lessen the impact on the human body by making devices smaller in size (or low-invasive). As such, in recent years, there has been an increasing need to make these devices more compact. This is why Murata developed and put onto the market medical grade multilayer ceramic capacitors that feature a compact size, large capacity and high reliability for implantable medical devices.
This has enabled the high-density design of implantable medical devices that has helped make devices even more compact in size. The GCH/GCR Series for implantable medical devices has been screened to ensure that the initial failure rate is lower than that of the MLCC for consumer devices. Also, it has been designed to have a longer service life. In humidity load tests, thermal shock cycles and other tests, the required specifications for MLCC for high-reliability devices are considerably different from those for MLCC for consumer devices. Although the circuits of implantable medical devices can be separated into life support and non-life support circuits, Murata recommends the GCR Series for life support circuits and the GCH Series for non-life support circuits, and makes the following recommendations for the following implantable devices.
Murata continues to extend the GCH Series lineup with compact and high-capacitance products that help make more compact implantable medical devices possible.
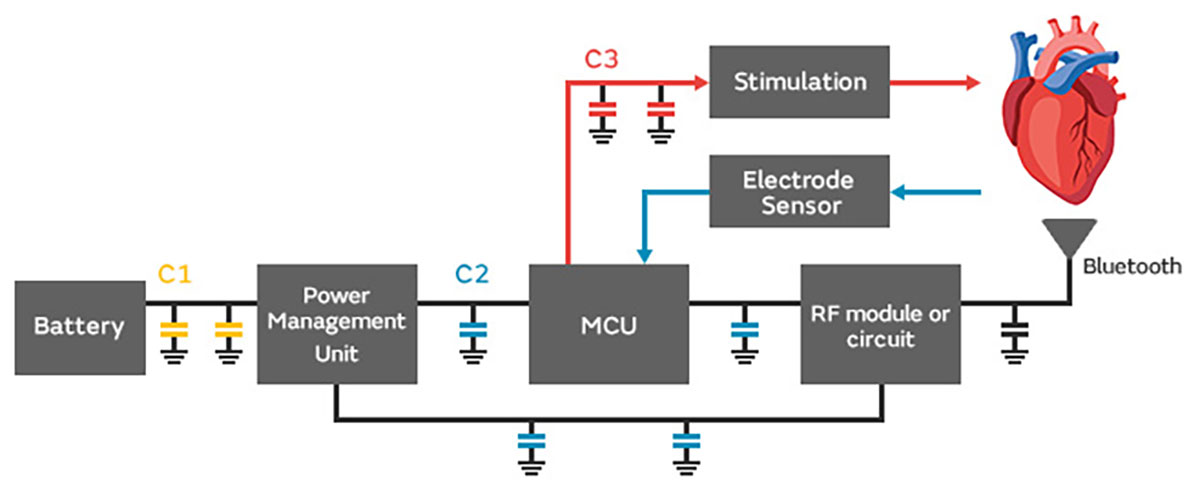
| C1 Energy storage for Battery | C2 decoupling for CPU | C3 Energy storage for stimulation | |
|---|---|---|---|
| Operating voltage | 1.8 to 3.6V | 0.9 to 1.2V | 10 to 20V |
| Rated voltage | 6.3V to 10V | 6.3V to 10V | 16V or 25V |
| Capacitance | 1 to 2.2uF | 10 to 47uF | 2.2 to 10uF |
| Example Recommendation | GCH188R70J225KE01# (0603/6.3V/2.2uF) GCH188C71A225KE01# (0603/10V/2.2uF) | GCH188R60J106ME11# (0603/6.3V/10uF) GCH188R61A106ME11# (0603/10V/10uF) | GCH31CR71C106KE01# (1206/16V/10uF) GCH188R61C475KE11# (0603/16V/4.7uF) |
The thin layer technology of the dielectric layer is an important point in the compactness and large capacity Murata strives for in our capacitors. We have established a processing technology to control the size and shape of the grains of the ceramic powder at high accuracy, and to distribute it equally at high density.
Thinner, smaller, and more accurate. Murata continues to create leading-edge capacitors.