Murata's Oxide-based Solid-state Batteries for Expanding the Range of Applications for IoT Devices and Supporting More Advanced Wearables (Part 2 of 2)
In Part 1, we explained how Murata Manufacturing Co., Ltd. ("Murata" below) developed a solid-state battery for practical applications that features both safety and high performance and that would be suitable for installation in IoT devices and wearables. With safety as the highest priority, Murata selected oxide-based ceramic material as the electrolyte*1 for developing practical applications and for enabling mass production of solid-state batteries, but sufficient performance could not be obtained using existing technology. In Part 2, we asked the engineers who were involved in development how they resolved challenging issues, the features of the completed solid-state battery, and the direction of future development using this battery and its technology.
*1: An electrolyte is a substance that serves as the path for charges and interchanging media (lithium ions in the case of a lithium-ion secondary battery) to move between the positive and negative electrodes inside secondary batteries which can charge and discharge power.
Development Roadmap Was Clearly Defined, but Difficult to Implement
--Although using oxide-based ceramic material as a solid electrolyte enables creation of a solid-state battery with high safety, a key point of frustration is that only batteries with low performance can be developed using conventional technology. What innovations could you come up with to overcome this technical hurdle?
To achieve satisfactory characteristics, we primarily used three approaches: development of solid electrolyte material with high ion conductivity, technology for forming precise, thin electrolyte layers, and development of processes for improving the adhesion of electrode active materials and electrolytes. Of these approaches, the formation of precise, thin electrolyte layers proved to be a challenging issue, but it was resolved by drawing on Murata's extensive technology and expertise for mass producing multilayer ceramic capacitors (MLCCs). Like solid-state batteries, MLCCs have a structure where the area between the electrodes is filled by a dielectric body made of ceramic material. A technology is available for creating a clean, thin ceramic film and then fully hardening, enabling mass production of microscopic pattern elements at high quality. We believed if this technology is used in the manufacturing of solid-state batteries, we could resolve the most difficult technical hurdle.
--When using the MLCC processes for Murata's solid-state batteries, were you able to resolve the issues as expected when the technology was first used?
To conduct this development, we assembled specialists in solid-state batteries and in MLCCs to launch the development project, but in reality, it turned out to be quite a challenge.
A firing process for integrating the sheet-shaped layered structures used in MLCC manufacturing was used to manufacture solid-state batteries. For MLCCs, products having more microscopic structures than solid-state batteries had been produced before without any problems, and so we thought that solid-state batteries could also be produced easily. However, firing was an extremely delicate process because battery performance will vary tremendously depending on the conditions, presenting us with significant struggles to produce a high-quality battery.
There was a fundamental cause for this. Although we were dealing with the same ceramic materials, the parameters that affect the performance and quality of the entire device are different between the dielectric body of the MLCC and the solid electrolytes of the solid-state battery. For this reason, the MLCC process had to be fine-tuned specifically for solid-state batteries.
Refining MLCC technology for manufacturing of solid-state batteries
--Do you mean that, even though the solid electrolytes that connect the positive and negative electrodes in a solid-state battery are the same ceramic material as the dielectric body inserted between the electrodes in an MLCC, in terms of their electrical role, they are a completely different thing? In situations where there were technical differences that can only be observed through testing, how did you resolve this problem?
We combined our MLCC manufacturing technology with our knowledge of batteries, and examined the materials, processes, and manufacturing equipment and their interactions with each other, and we made revisions until we obtained improvements. However, the specialists in solid-state batteries were not completely knowledgeable about MLCC manufacturing technology, and the specialists in MLCC did not fully understand the details about battery-specific behavior. And so, the members of the development project conducted thorough discussions, and through a repeated process of trial and error, they were able to find conditions enabling both high performance and quality for batteries for leading us to our solid-state battery.
Effects of achieving both high safety and high energy density
--You used Murata's proprietary MLCC manufacturing technology to successfully develop manufacturing technology for the future solid-state battery. What types of features does the newly-developed solid-state battery have?
We were able to obtain an energy density*2 that is 10x to 100x more than that of any previous oxide-based solid-state battery. For instance, even a compact battery with a size of 4 mm x 5 mm x 9 mm can provide an output of more than 10 mA, which is required for wireless transmission of data using Bluetooth LE. In our prototypes, we were able to verify maximum capacities of 20 to 30 mAh, and this level is sufficient to become a possible replacement for existing lithium-ion secondary batteries that are being used as the power supply for wireless earphones. In addition to that, because a design is used that obtains the same 3.8 V output voltage as existing lithium-ion secondary batteries, this battery will be easy to use when incorporated into electronic devices.
*2: The energy density is a measure of the amount of power that can be stored per unit volume or weight of a secondary battery. The units are Wh/L (volume-based units) or Wh/kg (weight-based units). A battery with a high energy density is capable of supplying a large amount of power despite having a compact size and lightweight design. Furthermore, this enables the creation of user-friendly mobile devices that have even more advanced electronic functions.
Previously, the conventional thought was that high capacities were difficult to achieve using oxide-based solid-state batteries. For this reason, the visitors at CEATEC 2019 who saw our prototype were astonished at how we achieved a fully-functioning battery with this high capacity and compact size.
Contributing to the realization of tough IoT devices and attractive wearables
--By focusing on achieving both a compact, lightweight design and a high capacity, you can help to drive the development of high added-value IoT devices and wearables.
The features of the completed solid-state battery do not stop at its high energy density. Because the oxide-based material that is used has a high thermal resistance, it also allows surface installation on PCBs subjected to high temperature environments due to operation or reflow*3.
*3: Reflow is a type of process for soldering electronic components to PCBs in the assembly line for electronic devices. The required electronic components are placed on a solder paste that was spread onto the PCB beforehand, and this is heated in an oven for melting the solder and providing a single connection between the board and electronic components.
Because operation is possible at high temperatures, this also enables IoT devices to be put in harsher environments. These batteries may also be suitable for joint usage with energy harvesting*4 technology for capturing light, temperature differences, vibrations, and other energy already found in the surrounding environment and converting it to power for use as an energy source. This is because devices that use energy harvesting are generally placed outdoors or in other harsh environmental conditions.
*4: Energy harvesting is a technology for capturing the already-existing energy in the surrounding environment where the powered electronic device is being used and converting it to power for use as a power source. This enables the device to supply its own power so that no battery changes or charging is required for producing a maintenance-free electronic device. The most popular energy source is light, and solar cells are used to convert to power. There have also been proposals for technology that uses road vibrations, force pressed on a switch, air temperature differences, and radio waves transmitted in the air as sources of energy. However, because the power that can be obtained on site is generally small and unstable, the battery must still be charged, and if necessary, a stable power supply must be provided.
Also, because assembly is possible using reflow, this eliminates the later process of mounting the battery only, enabling lower production costs for devices. In addition, the compact battery can be fitted into the empty spaces where components are not installed on the PCB for enabling an even smaller installation area.
For wearables which will be constantly worn and used, one requirement for their widespread use is the capability to use compartments with attractive designs. However, for previous batteries, the battery installation space had to be prepared beforehand, and so this limited the range of freedom for compartment designs. Newly developed batteries will give priority to the compartment design, and the battery can be placed within the gaps in the semiconductors and electronic components.
Optimized for a wide range of fields
--Looking forward, how is Murata planning to further develop and enhance the solid-state battery?
Actually, this technology that we have developed has a feature which will be particularly useful in further development of solid-state batteries. Most of the solid-state batteries that were developed before had a type of compatibility for the combination of materials used for the positive and negative electrodes and for the solid electrolyte. For this reason, materials could not be selected that matched the required characteristics. In contrast, Murata's technology enables a relatively wide selection for the materials that make up the battery. As a result, this allows manufacturers to change the output voltage or produce batteries with an emphasis on longevity or performance for easily making derivative versions of batteries.
Of course, development can also aim for batteries with more compact sizes and even higher performance. We also want to study the use of substances other than the currently-used lithium ion as media for storing the charge within a battery. When doing so, we will be studying solid electrolytes and other component materials from a completely new perspective.
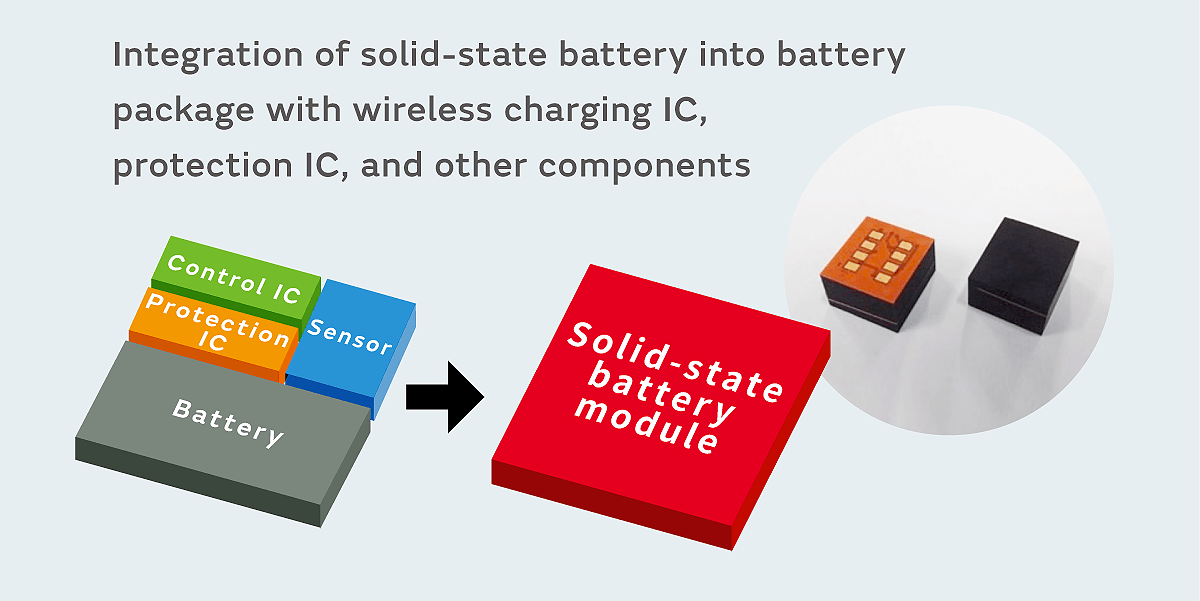
Murata also has technology for producing a wide range of electronic components besides batteries. If we use the high PCB installability of solid-state batteries, we will be able to produce battery modules integrated with battery control circuits, wireless power supply circuits, communication antennas, various sensors, and other components. This will enable us to provide solutions optimized for functionality and performance for a wide range of applications.

Dramatic expansion in usage applications for IoT devices and wearables
Murata has successfully developed a solid-state battery that will enable more compact sizes and higher capacities for mobile electronic devices while also ensuring high safety. This may also drive dramatic developments for wearables that remain constantly attached and for IoT devices that collect data in factories, road infrastructure, plants, and other harsh environments.
Murata's advancements in solid-state batteries have only just begun. The energy density of lithium-ion secondary batteries when they first came into the market in 1991 was only about one-third of the energy density of today's batteries. We will continue to research and improve these solid-state batteries that we have developed for achieving greater advancements in the future.
Other Links
Related articles
- Part 4: What are solid-state batteries? An expert explains the basics, how they differ from conventional batteries, and the possibility of practical application.
- Murata's Oxide-based Solid-state Batteries for Expanding the Range of Applications for IoT Devices and Supporting More Advanced Wearables (Part 1 of 2)

