We are looking for collaboration partners for Lithium-ion secondary battery characterization and degradation analysis.

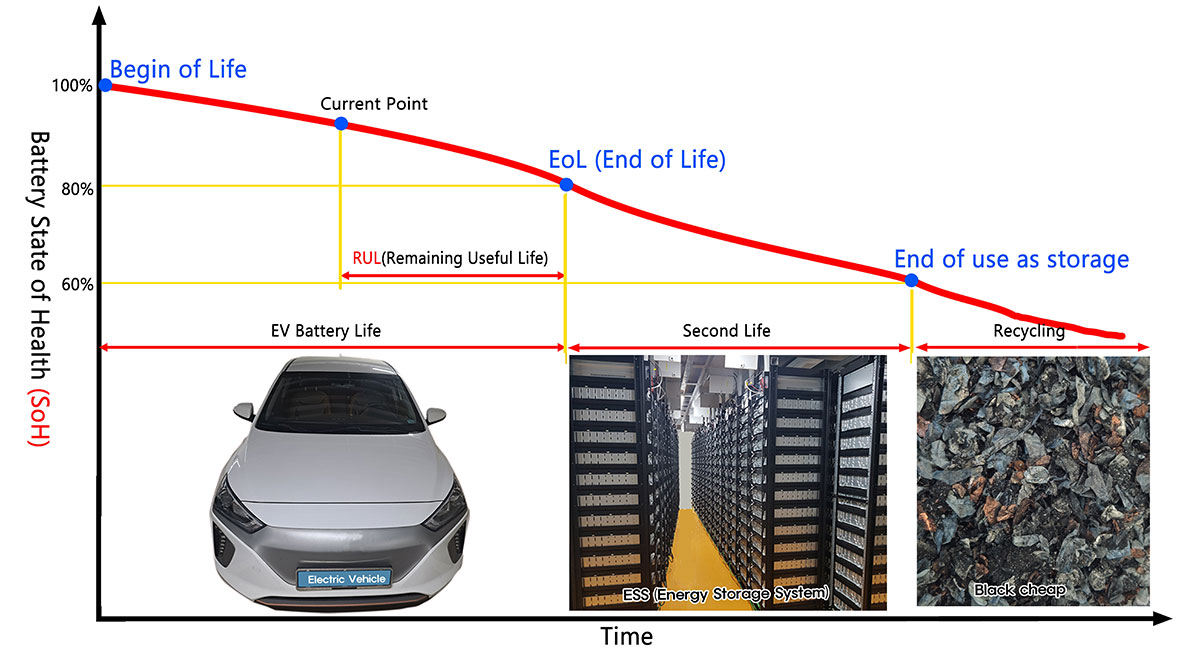
Lithium-ion battery (LiB) installation in electric vehicles (EV) and plug-in hybrid electric vehicles (PHEV) began to surge from around 2015, and these batteries are gradually reaching the end of their life. However, if used lithium-ion batteries are not disposed of properly, they may become a major environmental issue. Furthermore, to achieve a sustainable society, we must also promote the reuse of rare metals such as the lithium, cobalt, and nickel used in lithium-ion batteries.
In response to these issues, efforts to reuse, repurpose, and recycle lithium-ion batteries are attracting attention. In this article, we introduce lithium-ion battery characterization and degradation diagnosis, which are essential for these efforts.
Lithium-ion battery characterization and degradation diagnosis is a type of assessment that involves measuring lithium-ion battery characteristics such as battery capacity, internal resistance, and self-discharge rate, and diagnosing the battery degradation state.
Lithium-ion batteries are said to have a life of 2-3 years for those used in mobile devices such as smartphones, and 8-10 years or more for those used in electric vehicles (EV) and storage batteries, etc. However, this life varies depending on factors such as storage conditions, number of charge/discharge cycles, and operating temperature. Furthermore, in lithium-ion batteries comprising multiple cells, the degradation state of each cell may not be uniform.
It is difficult to determine the degradation state based solely on intuition during everyday use, such as charging and discharging. However, even in these cases, lithium-ion battery characterization and degradation diagnosis makes it possible to scientifically analyze the battery state and visually understand the degradation state with tables and graphs. This assessment is considered particularly important for the reuse, repurposing, and recycling of batteries.
There are three methods for reusing lithium-ion batteries that have become used up due to long-term storage or use: reuse, repurposing, and recycling. All are effective means of achieving environmental conservation and sustainability, but the methods of reuse differ. In this section, we explain the difference between these methods.
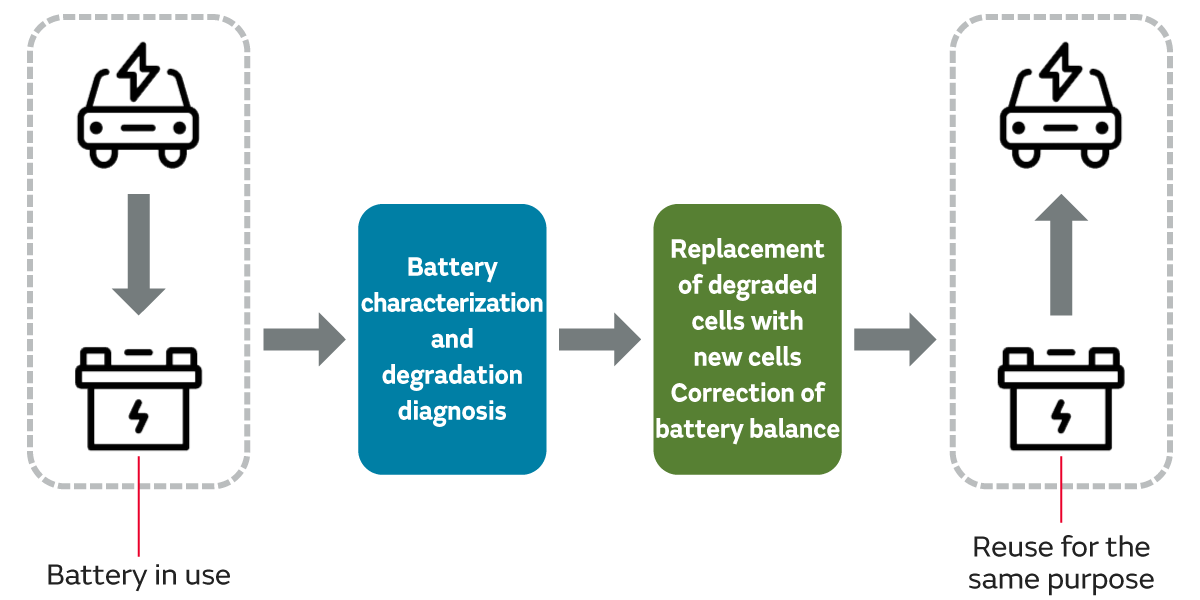
Reuse is a method of reusing used parts or products as they are, without manufacturing new items. Unlike repurposing, which is a recycling method that involves continued use for a different purpose, such as conversion from an automotive battery to a storage battery, reuse involves continued use for the same purpose. Specific examples for lithium-ion batteries include identifying degraded cells and replacing them with new ones, or correcting the battery balance, making it possible to use cells and modules for extended periods.
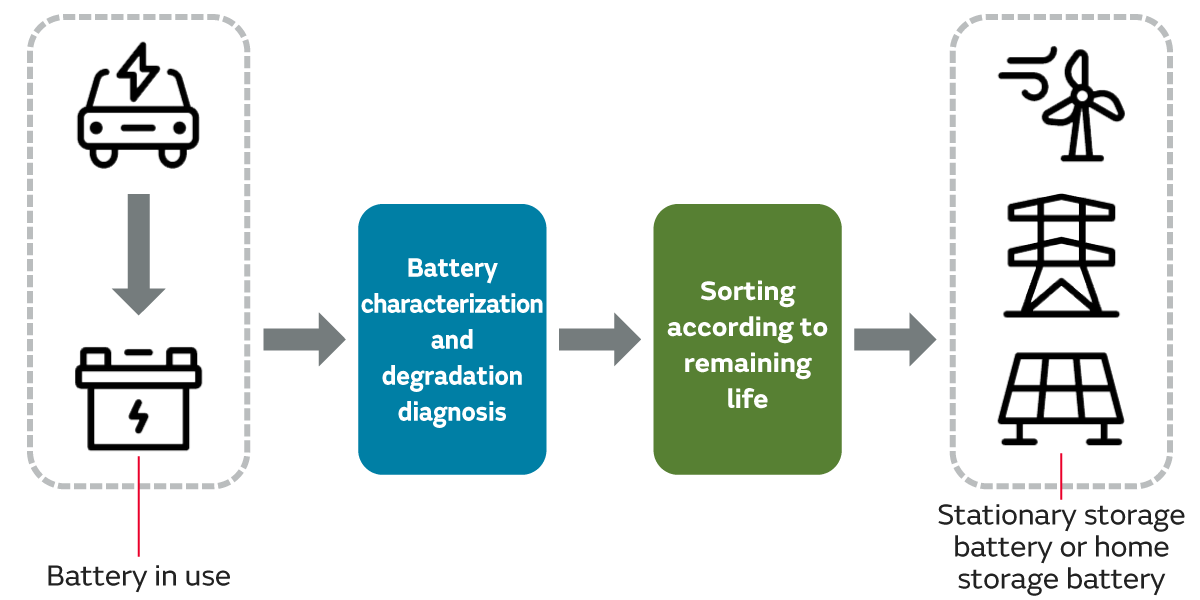
Repurposing refers to reusing batteries that have ended use in a product by incorporating them into a different product for a different purpose. Unlike recycling, which extracts just the raw materials from lithium-ion batteries that are no longer needed, repurposing enables the reduction of energy consumption and waste amounts by utilizing existing batteries as is for as long as possible.
A specific example is when high-performance automotive batteries, such as those used in electric vehicles, that have degraded after a certain period of use, are used as batteries for other purposes based on their remaining life, such as stationary storage batteries for factories or data centers, or as home storage batteries.
Recycling is a method of extracting reusable parts from waste or unwanted items, and reusing them as raw materials or energy. In the case of lithium-ion batteries, the cells are disassembled and incinerated in a vacuum heating furnace, and then chemical components such as nickel, cobalt, and other rare metals are recovered through processes including acid leaching, solvent extraction, and electrolysis.
If lithium-ion batteries are not properly disposed of, the hazardous substances contained in them can cause soil and water pollution. Furthermore, rare metals such as nickel and cobalt are not only difficult to obtain, but their mining places a great burden on the environment. Recycling of lithium-ion batteries is attracting attention as a method of reusing resources that reduces these environmental impacts.
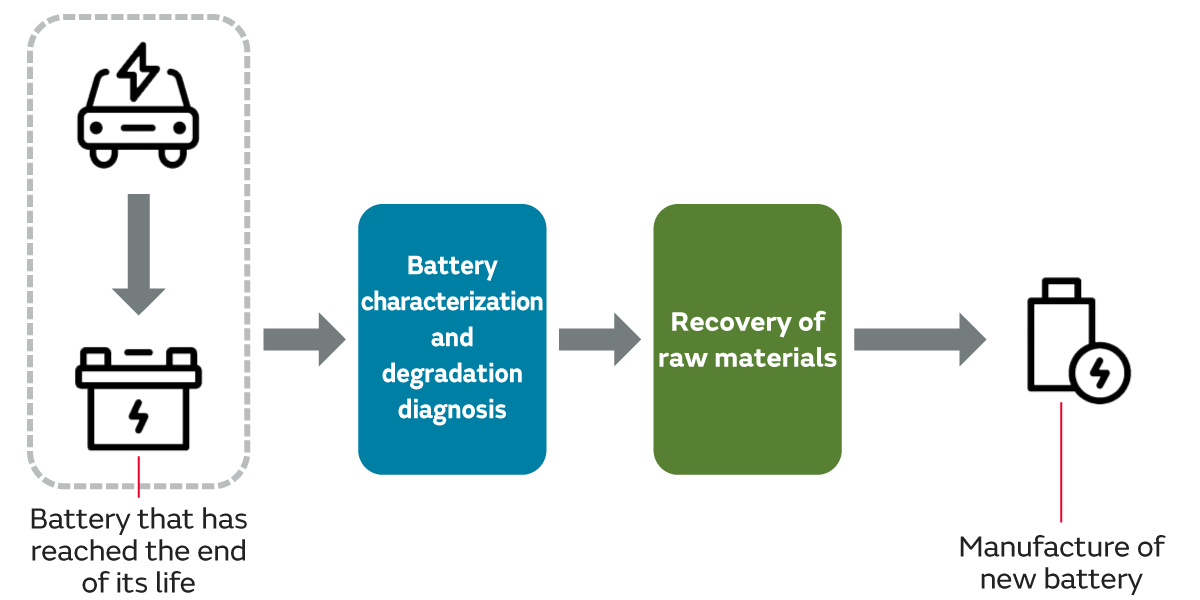
When reusing or repurposing a lithium-ion battery, the functionality and safety of the battery must be assured, and when recycling, it is necessary to confirm that the battery is no longer usable.
Lithium-ion battery characterization and degradation diagnosis is Murata's service for providing this assurance and confirmation. Lithium-ion battery characterization and degradation diagnosis makes it possible to visualize changes and degradation in battery characteristics due to aging, charging cycles, and operating and storage temperatures, making it possible to obtain data that is useful for determining battery life.
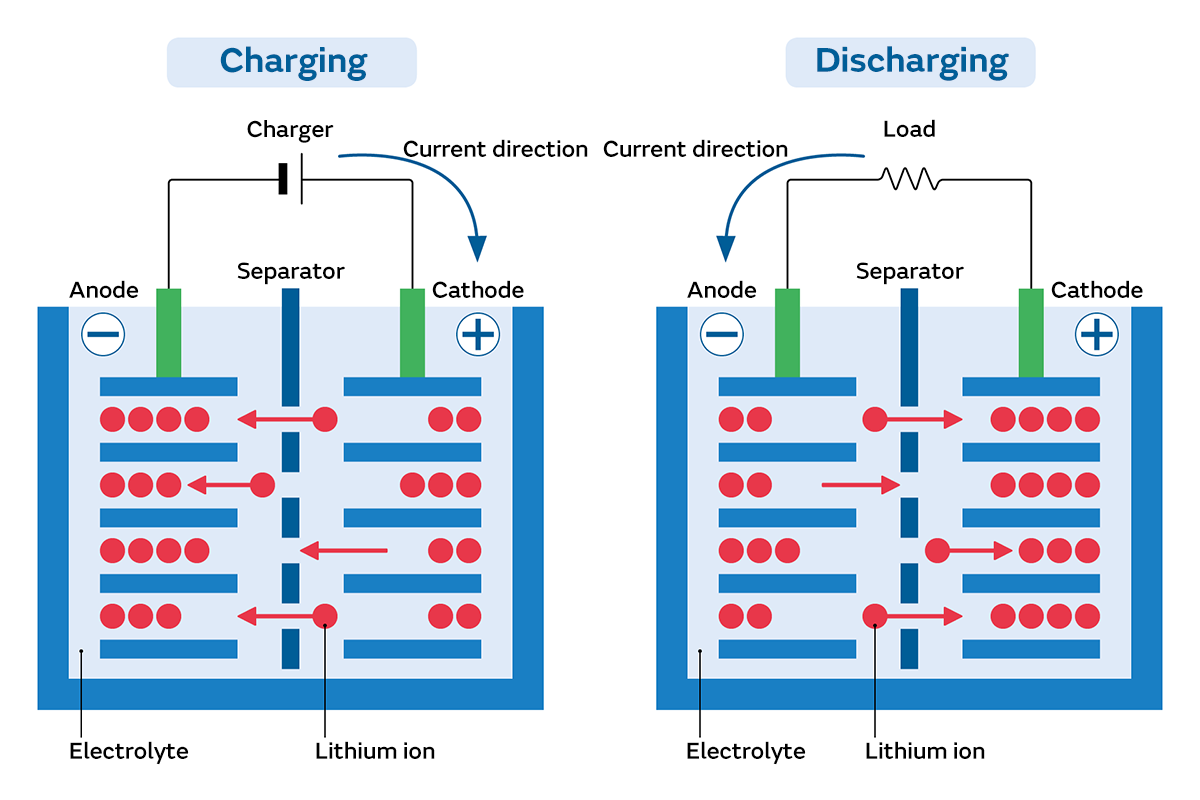
The greatest benefits of lithium-ion batteries are their high energy density and the ability to be charged repeatedly. However, as lithium-ion batteries are repeatedly charged and discharged, their performance gradually declines, symptoms of degradation appear such as the inability to charge fully or rapid charge depletion, and they eventually reach the end of their lives. What is happening inside a lithium-ion battery at this time? In this section, we explain the structural features of lithium-ion batteries, degradation phenomena, and the causes of degradation.
Lithium-ion batteries use a metal compound pre-loaded with lithium ions for the cathode, and materials such as carbon or lithium titanate for the anode that accumulates those lithium ions. The battery structure has a separator immersed in an electrolyte placed between the cathode and anode. This separator has a porous structure that allows lithium ions to pass through but prevents contact between the cathode and anode. This battery structure enables power generation without dissolving the electrodes in the electrolyte like conventional batteries. In addition, larger amounts of electricity can be stored compared to other batteries. Lithium ions move from the cathode to the anode during charging, and from the anode to the cathode during discharging. This means that even if the battery is discharged, it can be used repeatedly by charging it.
Lithium-ion batteries have the advantage of being capable of repeated use through charging and discharging. As use progresses, however, phenomena such as the inability to charge fully or rapid charge depletion occur, preventing them from performing as rated. Additionally, lithium-ion batteries may swell in some cases. These phenomena are referred to as degradation, and in the worst case may cause the battery to ignite or explode. Lithium-ion batteries that have degraded in this way are not only troublesome to use, but can also be dangerous. Therefore, in order to take advantage of the merits of lithium-ion batteries and continue use while avoiding risks, it is extremely important to understand the state of degradation.

Batteries generally degrade with use. Degradation phenomena can be broadly divided into "decrease in capacity" and "increase in resistance." In this section, we explain the mechanisms (principles) of these lithium-ion battery degradation types.
Degradation due to use includes cycle degradation, float degradation, and discharge degradation, etc.
Lithium ions move between the cathode and anode during charging and discharging. The capacity of a battery corresponds to the number of these lithium ions that move, and when the number of lithium ions that move (active lithium) decreases, the battery capacity decreases.
This decrease in capacity is expressed as a percentage called State of Health (SoH), which represents the ratio of the battery capacity after degradation (current capacity) compared to the state when the battery is new, which is set at 100%.
When the capacity decreases, phenomena such as the following have occurred inside the battery.
The types of degradation phenomena that occur vary depending on the battery design and how it is used. Additionally, there may be phenomena that reduce the safety of the battery, so caution is needed.
For example, if a side reaction causes the battery elements to swell and the internal structure becomes distorted, internal short circuits or other issues may also have occurred.
Batteries have something called internal resistance, which is the resistance inside the battery that hinders the flow of current. When current flows in a lithium-ion battery, the internal resistance generates heat, causing the voltage to drop. This heat generation and voltage drop significantly impact the efficiency and performance of lithium-ion batteries.
The internal resistance of lithium-ion batteries consists of ohmic resistance caused by the battery's conductive materials such as the electrodes and electrolyte, charge transfer resistance caused by electrochemical reactions in the charge/discharge cycle, and polarization resistance due to ion migration in the electrolyte.
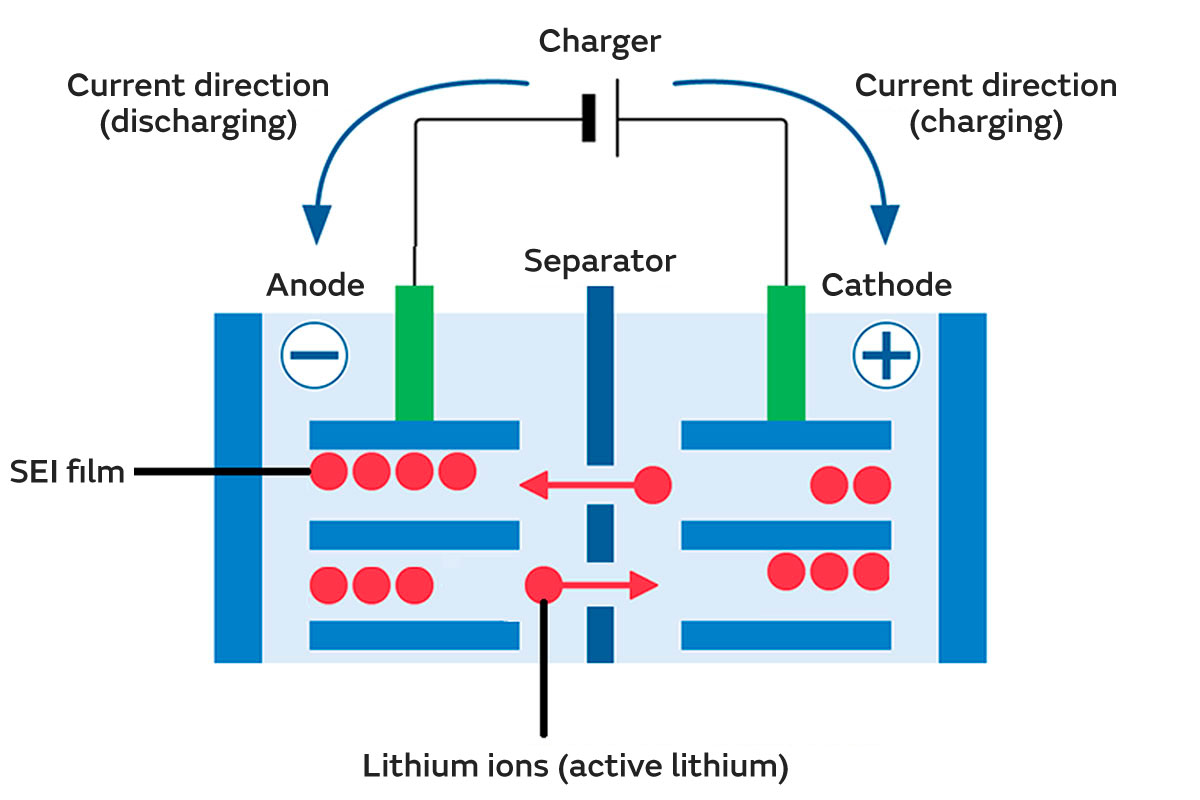
For example, during charging and discharging, SEI film is formed on the anode surfaces, which inhibits electrolyte reactions and decomposition. Ideally, this SEI film has an optimal chemical structure and thickness, but if the chemical structure of the SEI film is damaged or the film becomes excessively thick with use, this can increase the resistance.
If the internal resistance is high, more lithium ions than necessary must move inside the battery in order to supply the required power. This excess movement leads to reduced efficiency and performance. Additionally, high internal resistance produces a voltage drop that reduces the voltage applied to the load and can adversely affect the operation of devices.
There are many different methods for diagnosing the degradation state of lithium-ion batteries, but here we will explain four approaches: "History estimation method (data-driven SoH estimation)," "Resistance estimation method," "Charge/discharge method," and "Charge/discharge curve analysis method."
This method estimates the state of health (SoH) of a lithium-ion battery by obtaining the usage history data in real time. The SoH is estimated through a data-driven approach whereby machine learning is used to learn degradation patterns from large amounts of operation data and laboratory experiment data. This method has the advantage of being able to instantly understand the battery state, but also has the drawbacks that large-scale preprocessing is needed to identify characteristics strongly correlated with battery degradation, and the estimation accuracy varies widely depending on the data used. An additional issue is that it is applicable only to battery models for which learning data is available.
This method measures the internal resistance of a lithium-ion battery and estimates its degradation state. The internal resistance value is widely used as an indicator of battery characteristics, and lower internal resistance can be said to indicate better battery characteristics. The resistance estimation method has two methods for measuring the internal resistance: the AC method and the DC method.
This method applies an AC signal to the battery and analyzes the difference in the response speed of the battery's resistance components. This difference in response speed makes it possible to distinguish between reactive components resulting from electronic resistance or components resulting from the battery's redox reactions. Attempts are also being made to combine this method with data-driven approaches to analyze the resistance and also estimate the degree of decrease in the battery capacity based on the correlation between the AC response and how the capacity degrades. This method enables detailed analysis in a short time, but also has drawbacks such as that the responses vary depending on temperature and the remaining charge, and the high cost of a frequency response analyzer (FRA).
This method discharges the battery at a constant current, and calculates the internal resistance from the discharge current value and the voltage drop at a specific timing. This enables analysis using less expensive equipment compared to the AC method. This method includes various approaches such as analyzing the state with current flowing or analyzing the overvoltage relaxation after voltage application is stopped, enabling detailed analysis.
This test evaluates the SoH and other characteristics by charging and discharging with a specific method. It makes it possible to understand whether a lithium-ion battery can be used for a long period and how its performance changes.
Charging is generally performed by constant current-constant voltage (CC-CV) charging, which consists of first charging at a constant current, and then charging at a constant voltage once the battery voltage reaches a specified value (cut-off voltage). After charging, discharging is performed at a constant current until the specified voltage is reached.
This method estimates the state of a lithium-ion battery, such as its capacity and internal resistance, by analyzing the voltage changes during charging and discharging. The voltage changes during charging and discharging can be represented by a charge/discharge curve. Charge/discharge curves provide information about electrical characteristics such as battery capacity and internal resistance, and also about the lithium ions and other active materials inside the battery, making it possible to estimate the degradation state. For example, the same decrease in battery capacity may be due to a decrease in the cathode capacity or anode capacity, or due to a charging imbalance between the cathode and anode. Furthermore, the degree of decrease in the battery capacity differs depending on factors such as the rise in the resistance value. Charge/discharge curves contain information that indicates these battery internal states, and analysis of this information makes it possible to correctly understand the state of the battery.
Efforts are underway to develop alternative battery technologies to lithium-ion batteries, as well as batteries that do not use rare metals such as cobalt and nickel. However, use of lithium-ion batteries is expected to continue for quite some time going forward.
On the other hand, the extraction of rare metals entails significant costs and advanced technology, creating supply concerns relative to demand for lithium-ion batteries. Additionally, the disposal of large numbers of used lithium-ion batteries is becoming an environmental issue due to challenges involved in their processing. Reuse, repurposing, and recycling technologies are attracting attention as effective approaches for addressing these issues.
Murata Manufacturing has developed lithium-ion battery characterization and degradation diagnosis technology that addresses the technical and cost-related issues of conventional degradation analysis methods, and offers a service that enables easier understanding of battery states for parties involved in reuse, repurposing, and recycling.