Murata's Oxide-based Solid-state Batteries for Expanding the Range of Applications for IoT Devices and Supporting More Advanced Wearables (Part 1 of 2)
Aiming for Further Evolution of Mobile Electronic Devices
Due to tremendous advances in semiconductors and chip components, mobile electronic devices such as smartphones and notebook computers have become an essential part of our everyday lives and our work. Advancements in batteries for these devices have been a key driver in enabling use of these mobile electronic devices for a wider range of applications.
Before we can reach a fully data-driven society, mobile electronic devices must be designed in even more compact sizes and lightweight designs. To accurately assess the operating status of social infrastructure, factories, and other locations, compact IoT devices are essential for collecting on-site data and transmitting it to a data center. Wearables are also expected to become widespread for maintaining our health and enriching our lifestyles. However, further advancements in batteries are vital for realizing widespread use of these IoT devices and wearables.
Issues with High-Performance Lithium-ion Secondary Batteries
Currently, lithium-ion secondary batteries are the most widely used batteries for mobile electronic devices. Across the board, smartphones and similar devices use large amounts of power. Because they can store a large amount of power in a small space and can charge and discharge at high output, lithium-ion secondary batteries provide a high level of performance for mobile electronic devices far beyond any other types.
However, there are strong calls for even higher safety in today's lithium-ion secondary batteries so that they can be used in mobile electronic devices for even wider applications. The material components of lithium-ion secondary batteries include flammable liquids. Also, if a lithium-ion battery is subjected to an external impact, a short-circuit could occur within the battery, causing it to overheat, and in the worst case, it could even ignite. For this reason, lithium-ion batteries require various types of limitations and protective measures when using under harsh environments, in equipment critical to human life, or in equipment that handles valuable assets.
To resolve these issues, development is being conducted around the world on solid-state batteries where the electrolyte*1, which was a flammable liquid in lithium-ion batteries, is changed to a non-flammable solid material for enhanced safety. However, the solid electrolyte materials that have been used previously in solid-state batteries for high safety had a drawback where the ions did not flow easily within the battery, and this adversely affected the high performance of the lithium-ion secondary batteries. Development is being conducted on sulfide-based solid electrolyte materials which can provide high output, but when the sulfide-based material is exposed to air, toxic gas can be generated.
*1: An electrolyte is a substance that serves as the path for charges and interchanging media (lithium ions in the case of a lithium-ion secondary battery) to move between the positive and negative electrodes inside secondary batteries which can charge and discharge power.
Aiming for Solid-state Batteries Capable of Supporting Further Advances in IoT Devices and Wearables
At Murata Manufacturing Co., Ltd. ("Murata" below), we are developing solid-state batteries that boost the storable power to the maximum possible while ensuring safety as our utmost priority for contributing to the realization of compact, high-performance IoT devices and wearables. As a part of this effort, Murata is drawing on its long history of development in multilayer ceramic capacitor (MLCC) technology to develop proprietary materials, processes, and equipment technology.
In 2019, Murata successfully developed a solid-state battery that satisfied the highest industry performance standards. This prototype was unveiled at CEATEC 2019 and was awarded the CEATEC AWARDS 2019's Minister of Economy, Trade and Industry Award, and it has continued to attract growing attention.
Achieving Both Safety and High Performance by Combining Murata's Proprietary Materials Technology with MLCC Technology
The solid-state batteries developed by Murata are a groundbreaking technology that has the potential to expand the usage applications for mobile electronic devices. However, the road to this achievement has been filled with many challenges. We asked the Murata engineers who were involved in this development about how the development occurred, features of the developed solid-state battery, and the direction of advancements going forward.

Issue Is Combining Enhanced Safety with Improved Performance
--Currently, companies around the world are competing to develop a solid-state battery that will be the ultimate lithium-ion secondary battery. In this type of environment, what kind of solid-state battery is Murata trying to achieve?
We are focused on developing a solid-state battery with an energy density*2 suitable for use in IoT devices and wearables and where improved safety has the highest priority.
*2: The energy density is a measure of the amount of power that can be stored per unit volume or weight of a secondary battery. The units are Wh/L (volume-based units) or Wh/kg (weight-based units). A battery with a high energy density is capable of storing a large amount of power despite having a compact size and lightweight design. Furthermore, this enables the creation of user-friendly mobile devices that have even more advanced electronic functions.
When we think of where solid-state batteries will be used, for most people, the first thing that comes to mind is probably electric cars. However, the solid-state batteries being developed by Murata are a completely separate thing from those used in cars.
For car applications, for instance, to enable sudden acceleration of the car, sulfide-based materials are being widely studied because they provide superior ion conductivity for delivering output performance. However, sulfide-based materials present safety issues because toxic gas can be generated when the battery is damaged. The ensuring of high safety is also a key focus for wearables that people attach to themselves.

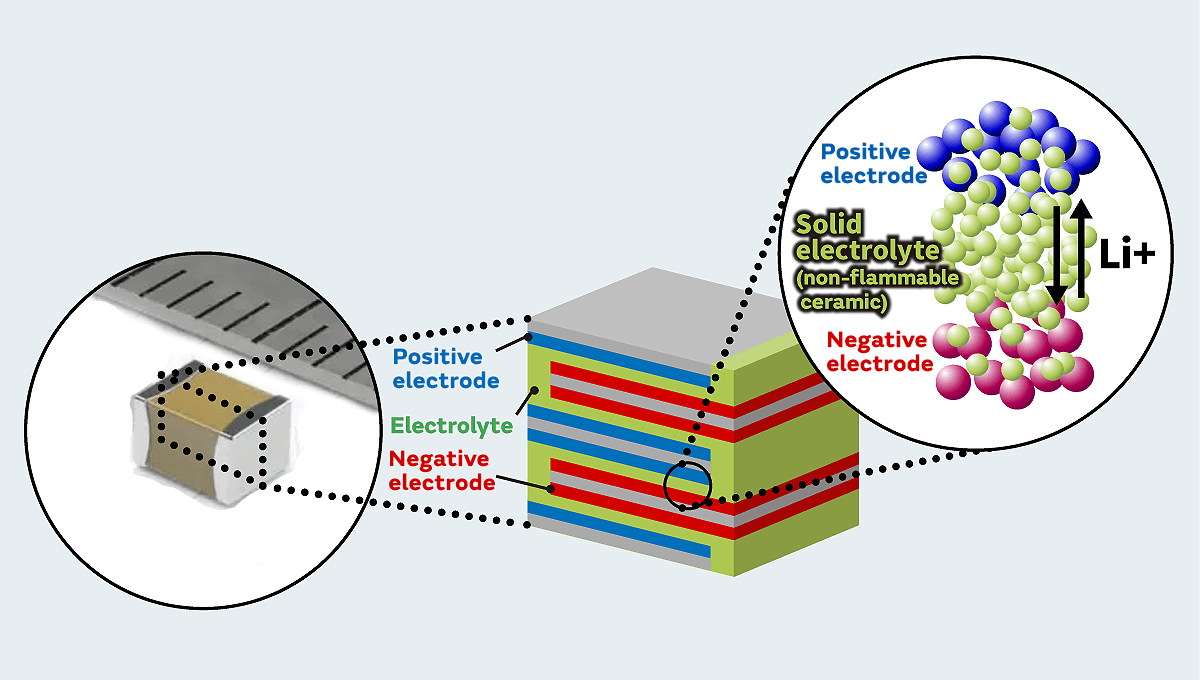
Because of these issues, Murata's solid-state batteries use a proprietary oxide-based ceramic material as a solid electrolyte because of its superior safety, thermal resistance, and non-flammable properties. Generally, the issues with oxide-based solid electrolytes have been how to improve the energy density and increase the capacity. IoT devices and wearables must have functions for collecting data from various sensors and transmitting the collected data wirelessly. Our objective was to achieve an energy density where the required power can be safely supplied for running these functions. To do this, we used materials developed by Murata to improve the energy density and increase the capacity.
MLCC Technology Was the Key for Achieving Both Safety and Performance for Solid-state Batteries
--Why is it difficult to improve the energy density when an oxide-based material is used as the solid electrolyte?
For all types of solid-state batteries, in order to create a battery with a high energy density, the ratio of the electrode active material*3 in the battery must be increased. Also, to achieve high output (low resistance), it is important to (1) improve the ion conductivity between electrodes and to (2) increase the interface adhesion between the electrode active material and electrolyte. This is not an issue with conventional lithium-ion secondary batteries because they use liquid electrolytes with high ion conductivity. However, even though the oxide-based ceramic material that we use has high safety performance, it has a relatively low ion conductivity and is a hard granular material, and this results in poor adhesion with the electrode active material. This is what makes it so difficult to attain both high energy density and high output.
*3: Active material refers to a substance making up the electrode whose role is to directly exchange charges.
- Continue reading: Murata's Oxide-based Solid-state Batteries for Expanding the Range of Applications for IoT Devices and Supporting More Advanced Wearables (Part 2 of 2)
Other Links
Related articles
- Part 4: What are solid-state batteries? An expert explains the basics, how they differ from conventional batteries, and the possibility of practical application.
- Murata's Oxide-based Solid-state Batteries for Expanding the Range of Applications for IoT Devices and Supporting More Advanced Wearables (Part 2 of 2)

